You often hear that “Europe bans 1,000+ cosmetic ingredients while the US bans only a handful”, which makes it sound like American products are wildly unsafe and European ones are automatically clean. The reality is more complicated.
1. Different legal architectures
The European Union uses Regulation (EC) 1223/2009 as its central cosmetic law, with annexes listing substances that are prohibited, restricted or explicitly allowed as colorants, preservatives and UV filters. Companies must appoint a Responsible Person within the EU, compile a Product Information File and ensure a cosmetic safety assessment has been performed before placing products on the market.
In the United States, cosmetics are regulated under the Federal Food, Drug and Cosmetic Act as updated by the Modernization of Cosmetics Regulation Act (MoCRA), which is phasing in stronger requirements for facility registration, product listing, safety substantiation and mandatory recall authority for unsafe products. The US does not maintain as long an explicit list of banned substances, but the FDA can still act against adulterated or misbranded cosmetics and remove dangerous products from sale.
2. What the “banned list” numbers hide
It’s true that the EU has a long annex of prohibited substances, while historically only a small number of cosmetic ingredients were formally banned in US law. However, many items on the EU’s prohibited list are industrial chemicals, impurities or substances that were never actually used in modern cosmetics and are unlikely to be found in well‑formulated products in any major market.
Conversely, US authorities can and do object to products containing hazardous contaminants, mislabelled drugs sold as cosmetics or ingredients used at unsafe levels even if those substances are not named in a short banned list. The headline comparison of “EU bans 1,300 ingredients, US bans 11” misses the shared core: both systems rely on safety assessments and enforcement, not on the absence or presence of a single long list.
3. How ingredients are actually assessed
In Europe, the SCCS publishes detailed Notes of Guidance describing how cosmetic ingredients and products should be tested and how margins of safety should be calculated from toxicology data and realistic exposure scenarios. For higher‑risk categories like preservatives, UV filters and hair dyes, ingredients are only permitted when an SCCS opinion concludes that they are safe under specified conditions of use.
In North America and globally, independent panels such as the Cosmetic Ingredient Review (CIR) assess many of the same ingredients, reviewing animal, in vitro and human data to decide whether they are safe as used in current formulations. Health Canada likewise states that cosmetic ingredients must not pose a risk to human health under normal use and maintains a “Hotlist” of ingredients that are restricted or effectively banned in Canadian cosmetics. In all cases, regulators focus on risk (dose and exposure) rather than just hazard (what a substance can do at very high levels).
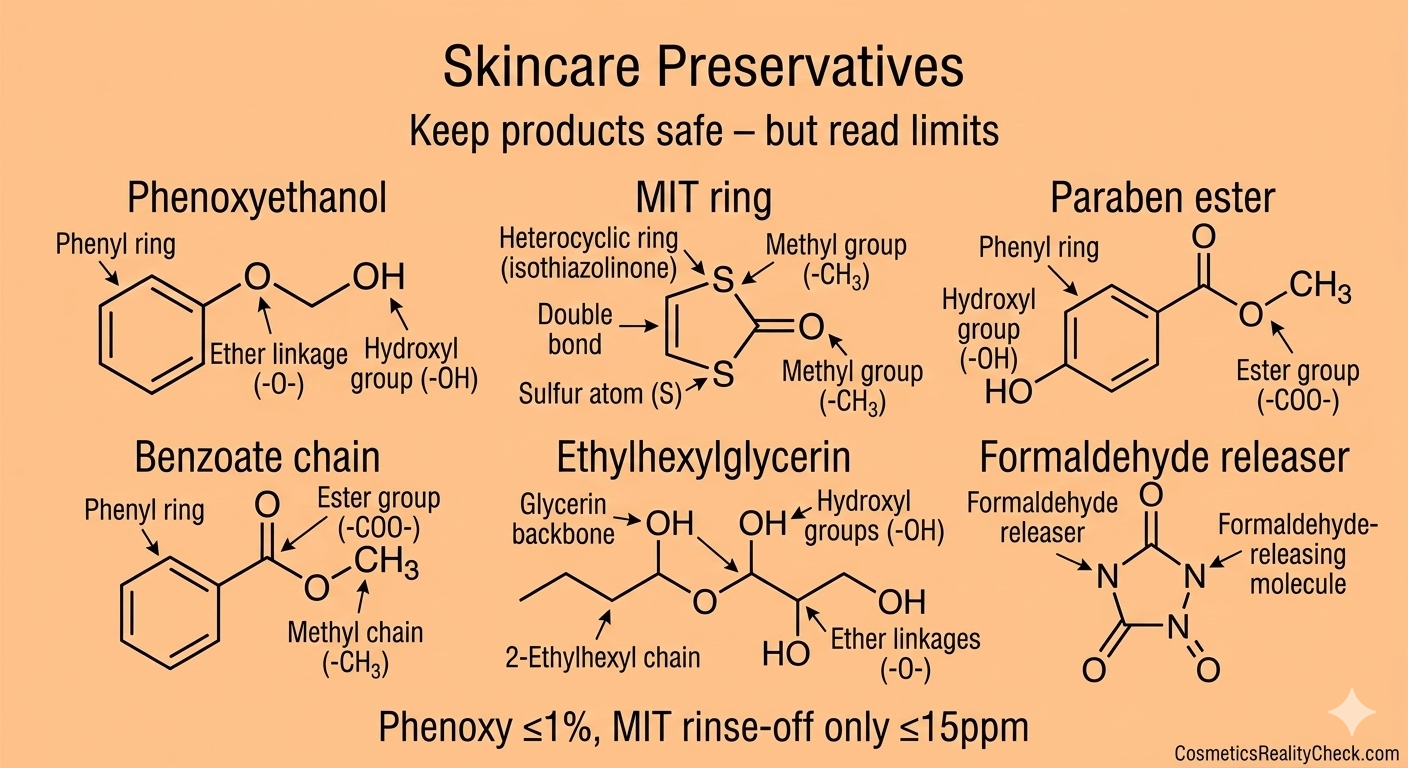
4. Where Europe is stricter – and where differences are shrinking
The EU has taken a more precautionary approach on several groups of substances, for example tightening or banning certain parabens, isothiazolinone preservatives and specific hair dye components earlier than other regions. It also requires more detailed labeling of fragrance allergens and imposes explicit maximum use levels for many UV filters and preservatives.
Recent US reforms under MoCRA, along with voluntary industry standards and global brand harmonization, are narrowing practical differences: large companies frequently align their formulas across markets rather than making one “safe EU” version and one “anything goes” US version. For most mainstream products, deviations tend to be in details (like allowed concentration of a specific preservative) rather than in an entire category being legal in one market and banned in another.
5. What this means for you as a consumer
European rules do create a somewhat tighter, more centralized pre‑market framework, while US regulation has historically relied more on post‑market enforcement and, now, gradually stronger up‑front requirements under MoCRA. But neither system guarantees that every product will be perfect for your skin; individuals can still react to ingredients that are considered safe for the general population, especially fragrance and certain preservatives.
Instead of assuming that all American products are unsafe or all European ones are harmless, it’s more useful to understand how safety is assessed, check whether an ingredient of concern has been reviewed by SCCS, CIR or your national authority and pay attention to your own history of irritation or allergy. Reading ingredient lists with this context in mind will help you make better decisions regardless of where a product was made.